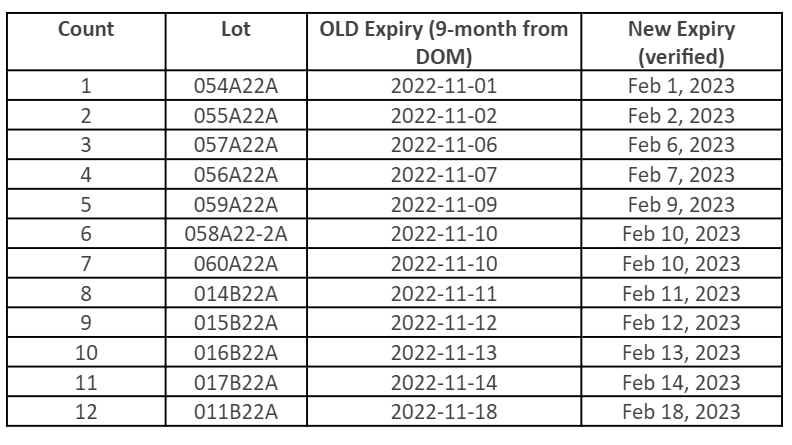
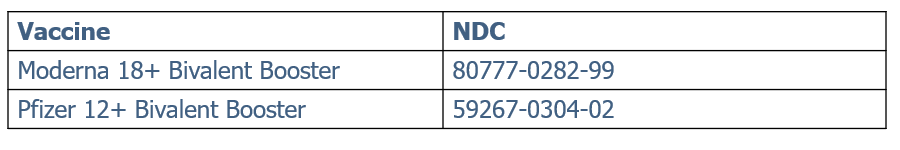
Limited Single Dose Vials of Pfizer Bivalent Becoming Available
Limited Single Dose Vials of Pfizer Bivalent Becoming Available
On Monday, November 14 through Tuesday, November 15, PDPH will open pre-ordering in Philavax for a limited amount of the updated (bivalent) Pfizer-BioNTech COVID-19 vaccine for people 12 years of age or older in single-dose vials. Deliveries will begin several days later. Single dose vials will not be available for ordering through Monday.com – partial ordering – or pickup.This limited rollout is intended to expand the number of locations that offer the updated vaccine. Only sites that would otherwise not be able to utilize the standard six-dose per vial bivalent Pfizer will be approved to order single dose vials. Large health systems and sites with regular patient volumes/throughput should continue to order the six-dose per vial bivalent Pfizer.Ancillary kits will not be distributed with orders of single-dose vials. Single-dose vials do not require the use of low-dead-volume (LDV) syringes.Quick Facts:Single-Dose Vial Pfizer Bivalent Booster (Ages 12+)• NDC: 59267-1404-02• Configuration: 10 single-dose vials/carton• Minimum quantity per order: 50 doses• Maximum quantity per order: 150 doses• Product dimensions: 1.457 in length × 1.535 in width × 3.504 in height• Storage and handling: Same as other Pfizer tris products (e.g., store at ultra-low temperature until expiry, may refrigerate up to 10 weeks within expiry period)• No ancillary kits included with single-dose vial orders